When you pick up a bottle of generic medication at your local pharmacy, it looks like a simple transaction. You hand over cash or insurance, and the pharmacist hands you a pill. But behind that counter lies one of the most complex, globalized, and financially intricate networks in modern healthcare. The generic drug supply chain is the network through which affordable medications travel from raw material sourcing to patient dispensation involves multiple stakeholders, international borders, and strict regulatory hurdles.
Unlike brand-name drugs, where the manufacturer captures the majority of the revenue, generic manufacturers often see only a fraction of the final cost. Understanding how these medicines move from factory floors in Asia to shelves in Bristol or Boston is key to understanding why prices fluctuate, shortages happen, and why your copay might change overnight.
The Journey Begins: Sourcing Active Pharmaceutical Ingredients
Every generic drug starts with an Active Pharmaceutical Ingredient (API) is the biologically active component of a medication responsible for its therapeutic effect. This is the actual medicine inside the tablet or capsule. The rest is just filler-binders, coatings, and dyes that make the pill stable and swallowable.
Here is the catch: very little of this critical ingredient is made locally. According to a 2021 study published in PMC, approximately 88 percent of API manufacturing occurs outside the United States. China and India dominate this sector, producing the vast majority of the world’s generic building blocks. Only about 12 percent of API production happens within U.S. borders.
This globalization creates efficiency but also vulnerability. When geopolitical tensions rise, or a factory in Gujarat shuts down for maintenance, ripples are felt instantly in pharmacies across Europe and North America. During the COVID-19 pandemic, this fragility was exposed when API shortages affected nearly 170 generic medications, according to FDA data. Manufacturers must navigate customs, quality inspections, and long shipping times before they even begin mixing their first batch.
Regulatory Hurdles: From ANDA to GMP Compliance
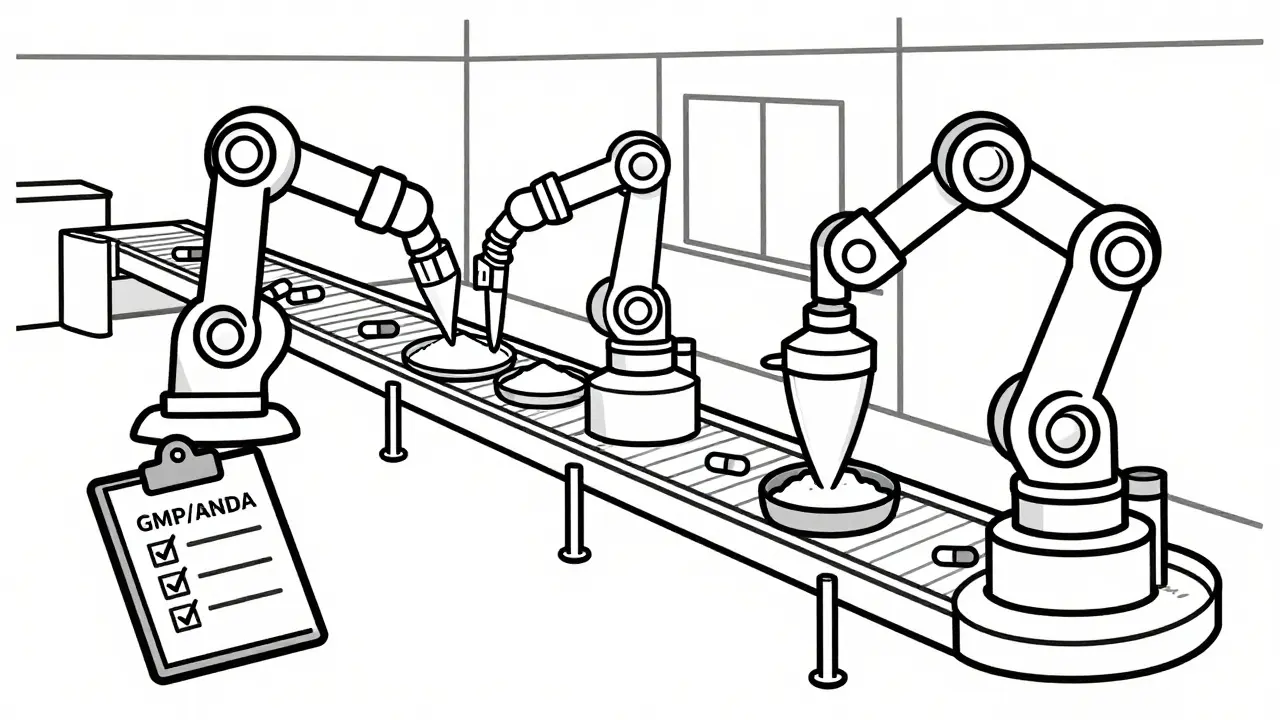
Once the API arrives, the manufacturer cannot simply start selling pills. They must prove that their generic version is therapeutically equivalent to the original brand-name drug. In the U.S., this requires filing an Abbreviated New Drug Application (ANDA) is a submission to the FDA demonstrating that a generic drug is bioequivalent to its branded reference product. This process, established by the Hatch-Waxman Act of 1984, allows generics to skip expensive clinical trials if they can prove identical chemical composition and absorption rates.
After approval, production begins under strict Good Manufacturing Practices (GMP) are regulatory standards ensuring consistent quality and safety in pharmaceutical production. These rules dictate everything from air filtration in clean rooms to the calibration of weighing scales. Quality control testing happens at multiple stages. If a batch fails, it is destroyed. No shortcuts are allowed because the stakes involve human lives.
Dr. David Ridley, a former FDA official, has noted that monitoring overseas facilities remains a significant challenge. Regulators struggle to keep pace with the volume of foreign sites, leading to increased inspection efforts. The FDA increased foreign facility inspections from 248 in 2010 to 641 in 2022, reflecting the growing complexity of this global web.
Distribution: Wholesalers and the Bulk Game
Once manufactured and approved, the drugs leave the factory and enter the distribution phase. This is where wholesale distributors are intermediaries that purchase drugs in bulk from manufacturers and sell them to pharmacies come into play. Companies like McKesson, AmerisourceBergen, and Cardinal Health act as the middlemen, buying millions of units at once.
These wholesalers negotiate hard. They often secure 'prompt payment discounts' for paying invoices early. According to the Association for Accessible Medicines’ 2024 supply chain brief, these discounts are negotiated on a case-by-case basis. The wholesaler then sells the drugs to pharmacies at a contracted discount off the Wholesale Acquisition Cost (WAC). The size of the pharmacy matters here. Large chains have more leverage to negotiate better rates than independent community pharmacies.
This step is crucial for inventory management. Pharmacies need to maintain ample stock to avoid running out of essential medications. However, holding too much inventory ties up capital and risks expiration. It is a delicate balancing act between availability and financial health.
The Financial Maze: PBMs, MACs, and Reimbursement
Perhaps the most confusing part of the supply chain is the financial layer involving Pharmacy Benefit Managers (PBMs) are entities that manage prescription drug benefits for insurers, processing claims and negotiating rebates. Three major players-CVS Caremark, OptumRX, and Express Scripts-control approximately 80 percent of the PBM market. They act as gatekeepers, deciding which drugs are covered and how much pharmacies get paid.
For generic drugs, reimbursement typically uses a Maximum Allowable Cost (MAC) is a ceiling price set by payers for reimbursing specific generic drug strengths and dosage forms methodology. Unlike brand drugs, which are reimbursed based on a percentage of a published list price, MAC sets a hard cap. For example, a payer might decide that any 10 mg tablet of atorvastatin will be reimbursed at $5.00, regardless of what the pharmacy paid for it.
This creates a risky environment for pharmacists. If the MAC price drops below the pharmacy’s acquisition cost, they lose money on every fill. A 2023 survey by the American Pharmacists Association found that 68 percent of independent pharmacy owners cited MAC pricing below acquisition cost as a major financial pressure. This disparity stems from structural differences in the supply chain. Generic manufacturers capture only 36 percent of the expenditure on generic drugs, compared to 76 percent for brand manufacturers, according to the University of Southern California’s Schaeffer Center.
Comparison: Generic vs. Brand Supply Chains
| Feature | Generic Drugs | Brand-Name Drugs |
|---|---|---|
| Manufacturer Revenue Share | 36% of total expenditure | 76% of total expenditure |
| Reimbursement Method | Maximum Allowable Cost (MAC) | Percentage of List Price |
| Pricing Leverage | Low; intense competition | High; patent protection |
| Rebate Negotiations | Rarely engaged with PBMs | Frequent; substantial leverage |
| API Sourcing | 88% sourced internationally | Varies; often domestic or diversified |
As the table shows, the economics are vastly different. Generic manufacturers face a 'race to the bottom' in pricing. With hundreds of competitors making the same molecule, there is little room for profit margins. This intensity drives innovation in logistics but also increases the risk of shortages if a manufacturer decides a drug is no longer profitable to produce.
Future Trends: AI, Blockchain, and Resilience
The industry is not standing still. To combat vulnerabilities, companies are adopting new technologies. AI-driven demand forecasting is using machine learning algorithms to predict drug demand and prevent shortages helps manufacturers adjust production schedules before a shortage hits. By analyzing real-world data (RWD), companies can pinpoint bottlenecks and respond to delays faster than ever before.
Blockchain is also emerging as a tool for transparency. By creating an immutable ledger of every step in the supply chain, stakeholders can verify the authenticity and condition of drugs. This is particularly important for combating counterfeit medications, which remain a global threat.
Furthermore, there is a push for diversified sourcing. Relying on a single country for APIs is seen as a strategic risk. Governments and manufacturers are exploring options to bring some production back home or spread it across more regions. The FDA’s 2023 Drug Competition Action Plan includes initiatives to streamline approvals and address shortages through enhanced communication with manufacturers.
Why This Matters to You
You might wonder why you should care about API factories in India or PBM negotiations in Pennsylvania. The answer is simple: access and affordability. The current system delivers 90 percent of all prescriptions filled in the U.S., yet represents only 23 percent of total drug spending. It works, but it is fragile.
When you see a drug shortage notice in your pharmacy, it is often the result of a break in this complex chain-a delayed shipment, a failed quality test, or a pricing dispute that made production unviable. Understanding the supply chain helps explain why switching brands sometimes isn't an option, or why your pharmacist might call you to discuss alternatives. It transforms a mysterious administrative hurdle into a logical, albeit imperfect, system designed to keep you healthy.
Who makes generic drugs?
Generic drugs are made by specialized pharmaceutical manufacturers who have received approval from regulatory bodies like the FDA. While many large companies exist, the top 10 generic manufacturers control about 65 percent of the U.S. market. Most of the raw materials (APIs) used by these manufacturers are produced in China and India.
Why are generic drugs cheaper than brand names?
Generics are cheaper primarily because manufacturers do not incur the high costs of initial research, development, and clinical trials. They also benefit from intense market competition, as multiple companies can produce the same molecule once patents expire. Additionally, the supply chain structure allows for lower marketing and distribution costs compared to brand-name drugs.
What is a PBM and why do they matter?
A Pharmacy Benefit Manager (PBM) is an intermediary that manages prescription drug benefits for insurance companies. They process claims, determine which drugs are covered, and negotiate prices with manufacturers and pharmacies. PBMs play a critical role in setting reimbursement rates, such as Maximum Allowable Costs (MAC), which directly impact how much pharmacies earn per prescription.
How does the supply chain affect drug shortages?
The generic supply chain is highly sensitive to disruptions. Since most APIs are manufactured overseas, issues like transportation delays, regulatory holds, or factory shutdowns can halt production. Furthermore, thin profit margins mean manufacturers may stop producing low-demand drugs, leading to shortages. The lack of redundancy in the supply chain exacerbates these issues.
Is the generic drug supply chain safe?
Yes, the supply chain is rigorously regulated. Manufacturers must adhere to Good Manufacturing Practices (GMP) and pass FDA inspections. While global sourcing presents challenges, regulatory agencies have increased oversight and inspections of foreign facilities to ensure quality and safety. Technologies like blockchain are also being adopted to enhance traceability and prevent counterfeit products.