When you hear that a new drug has a 20-year patent, it’s easy to assume the company has two decades to sell it without competition. But in reality, most drugs have less than half that time on the market. The truth is, effective patent life-the actual window of market exclusivity-is often just 10 to 15 years, and sometimes even less. Why? Because the clock starts ticking long before the drug even reaches pharmacy shelves.
Patents Start Before the Drug Is Even Tested
Under U.S. law, a pharmaceutical patent lasts exactly 20 years from the date it’s filed. Sounds straightforward, right? But here’s the catch: companies file patents during early research, often before human trials begin. That means years of development-preclinical testing, Phase 1, 2, and 3 clinical trials, plus FDA review-eat up a huge chunk of that 20-year term before the drug is even approved for sale.On average, it takes between five and ten years to get a new drug from lab to pharmacy. That’s not a guess. Data from Drug Patent Watch and the American Journal of Managed Care shows that by the time a drug gets FDA approval, anywhere from 50% to 70% of its patent life is already gone. So if a patent was filed in 2010, and the drug hits the market in 2018, the company only has 12 years left before generics can arrive-not 20.
The Hatch-Waxman Act: A Deal With Trade-Offs
In 1984, Congress passed the Drug Price Competition and Patent Term Restoration Act, better known as the Hatch-Waxman Act. Its goal? Balance two competing interests: reward innovation and let generics in quickly to lower prices. To do that, it created a system where patent holders could get back some of the time lost during regulatory review.This is called Patent Term Extension (PTE). It lets companies extend their patent by up to five years-but with strict rules. First, the extension can’t push the total market exclusivity past 14 years after FDA approval. Second, it only applies to the original patent covering the active ingredient, not later ones. And third, the drug can’t have been approved before. That last rule blocks companies from getting extensions for minor tweaks to old drugs.
Still, even with PTE, the math rarely adds up to 20 years of protection. Most drugs end up with 12 to 15 years of effective exclusivity. For some, it’s closer to 10.
Secondary Patents: The Hidden Game
Here’s where things get tricky. While the original patent might expire in 12 years, companies don’t just sit back. They file dozens of follow-up patents-on things like new dosages, pill coatings, delivery systems, or combinations with other drugs. These are called secondary patents, and they’re not covered by Hatch-Waxman’s extension rules.A 2023 study by the R Street Institute looked at 432 new drugs approved between 1985 and 2005. The average drug had 17 patents tied to it. Blockbuster drugs? Some had 20 to 30. These aren’t always about better medicine. Many are designed purely to delay generics. A drug might get a patent on an extended-release version, or on a specific metabolite, or on a new combination therapy. None of these are new active ingredients, but they’re enough to trigger lawsuits and delay generic entry.
And it works. The study found that higher-selling drugs are 37% more likely to get these secondary patents. The most profitable drugs get the most legal shields. This practice is called “evergreening,” and it’s become standard in the industry. The FDA lists all these patents in the Orange Book, making it easy for brand companies to sue generic manufacturers the moment they file for approval.
The 30-Month Stay: A Legal Delay Tactic
When a generic company files an application with the FDA, it must notify the patent holder. If the brand company sues within 45 days, the FDA can’t approve the generic for 30 months. That’s not a coincidence-it’s a built-in delay.This 30-month stay is meant to give courts time to decide if the patent is valid. But in practice, it’s often used as a strategic tool. Even if the patent is weak, the lawsuit alone buys time. Many generic companies can’t afford to fight multiple lawsuits, so they wait. Some never enter the market at all.
And here’s the kicker: even after the 30 months are up, if the court hasn’t ruled yet, the FDA still can’t approve the generic. The process drags on. Companies use this to stretch exclusivity well beyond the original patent term.
Regulatory Exclusivities: Extra Layers of Protection
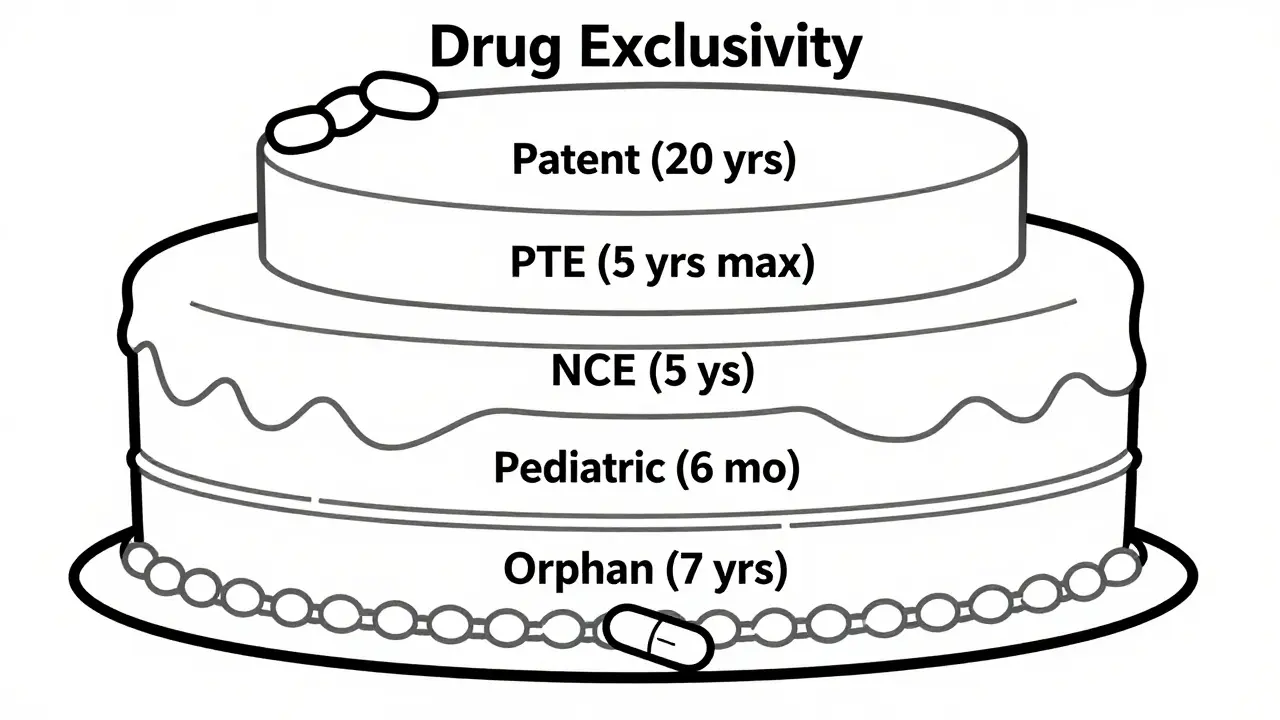
Patents aren’t the only tool. The FDA also grants regulatory exclusivities-separate from patents-that block generics even if the patent has expired.- New Chemical Entity (NCE) Exclusivity: 5 years of protection for a completely new active ingredient.
- New Clinical Investigation Exclusivity: 3 years for new uses or formulations of existing drugs.
- Orphan Drug Exclusivity: 7 years for drugs treating rare diseases (under 200,000 U.S. patients).
- Pediatric Exclusivity: 6-month extension if a company studies the drug in children.
These don’t extend patents. They’re independent. So a drug might have a patent expire in 2027, but still be protected until 2030 because of NCE exclusivity. And if it gets pediatric exclusivity? That pushes it to 2030.5. These layers stack on top of each other, making it harder for generics to break in.
Global Differences: It’s Not Just the U.S.
The U.S. system isn’t unique. Other countries have similar, but different, rules.- In Canada, companies can apply for a Certificate of Supplementary Protection (CSP), which adds up to 24 months after patent expiry.
- Japan allows up to 5 years of patent term extension for regulatory delays-similar to the U.S., but with different calculation rules.
- The European Union has Supplementary Protection Certificates (SPCs), which can extend protection by up to 5 years, with an additional 6 months for pediatric studies.
But even with these extensions, the pattern is the same: the clock starts early, and the real market life is always shorter than the paper patent.
The Economic Reality: Billions at Stake
Developing a new drug costs an estimated $2.6 billion (in 2013 dollars, adjusted for inflation, that’s closer to $3.5 billion today). That’s why companies need exclusivity. Without it, they can’t recoup their investment. But when exclusivity ends, prices crash. Generics can undercut branded drugs by 80% to 90% within a year.That’s why companies plan years ahead. When a drug’s exclusivity is about to expire, they shift marketing, pricing, and even R&D focus. Some launch their own generic versions. Others create “authorized generics” to keep some revenue. Others pivot to new indications or delivery methods to keep the product alive.
By 2025, global sales of drugs losing exclusivity are projected to hit $250 billion annually. That’s not just a number-it’s billions in revenue vanishing overnight for companies that didn’t prepare.
Is the System Working?
The Hatch-Waxman Act was designed to balance innovation and access. But today, the system is stretched thin. While original patents and extensions still play a role, the real drivers of long exclusivity are secondary patents and regulatory exclusivities. And they’re being used aggressively-especially on high-revenue drugs.Studies show 91% of drugs that get patent term extensions still maintain monopolies well past their expiration, thanks to these other tools. That’s not what Congress intended. The goal was to give companies enough time to recover costs-not to create permanent monopolies through legal loopholes.
Regulators are starting to push back. Courts are now more willing to strike down weak secondary patents. The FTC and Congress have held hearings on evergreening. But change is slow. For now, the system still favors companies that know how to play the game.
What This Means for Patients and Providers
For patients, longer exclusivity means higher prices. For prescribers, it means fewer affordable options. For insurers and pharmacies, it means unpredictable formulary changes and sudden spikes in spending.When a drug’s exclusivity ends, the market shifts fast. Pharmacists see a flood of generic requests. Clinicians scramble to switch patients. And patients pay less-but only after months or years of waiting.
The system isn’t broken. It’s working exactly as designed. But the design has been gamed. The 20-year patent was never meant to be the full story. The real story is what happens after the patent is filed-and that’s where the game is played.
Why isn’t the patent term extended to cover the full time spent in clinical trials?
The law does allow extensions, but they’re capped. Under the Hatch-Waxman Act, the maximum extension is five years, and the total market exclusivity can’t exceed 14 years after FDA approval. This was a deliberate compromise: enough time to recover costs, but not so much that generics are blocked for decades. The system assumes that most drugs will be approved within 10 years of filing. When it takes longer, companies get a partial extension-but not enough to fully restore the lost time.
Can a drug have more than one patent?
Yes. A single drug can have dozens of patents. The original patent covers the active ingredient. Then come secondary patents: on formulations, dosages, delivery systems, metabolites, or combinations with other drugs. These are filed after approval and aren’t subject to the same limits as the original patent. This is how companies extend exclusivity beyond the original 20-year term.
Do generic companies always wait for patents to expire before entering the market?
No. Generic manufacturers often challenge patents before they expire. They file Abbreviated New Drug Applications (ANDAs) and declare that the patent is invalid, unenforceable, or won’t be infringed. If the brand company sues, it triggers the 30-month stay. But if no lawsuit is filed-or if the court rules in favor of the generic-the FDA can approve the drug before the patent expires.
What’s the difference between a patent and regulatory exclusivity?
Patents are granted by the U.S. Patent and Trademark Office and protect inventions. Regulatory exclusivity is granted by the FDA and protects market access based on approval timelines. A patent can expire while exclusivity remains active. For example, a drug might lose patent protection in 2025 but still be protected by NCE exclusivity until 2030. Generics can’t enter until both expire.
Why do blockbuster drugs have more patents than low-selling ones?
Because the financial payoff is much higher. A blockbuster drug can generate billions in sales. Even a one-year delay in generic entry can mean hundreds of millions in extra revenue. Companies invest heavily in patent strategies for these drugs-filing multiple secondary patents, using legal tactics to delay generics, and extending exclusivity through new formulations. Lower-revenue drugs don’t justify the same level of legal effort.

8 Comments
Raman Kapri
March 2 2026
The article makes a compelling case, but it ignores the fundamental asymmetry in the system: while innovators are forced to file patents years before clinical trials, generic manufacturers face no such constraints. They wait, watch, and then strike with surgical precision the moment exclusivity wavers. This isn’t a flaw-it’s a feature of a market designed to extract value from innovation, not reward it.
Megan Nayak
March 2 2026
Let’s be real-this whole system is a theater of the absurd. We’re told we’re protecting innovation, but what we’re really protecting is the right of a handful of executives to siphon billions from sick people who can’t afford to wait. The 20-year patent? A fairy tale. The 12-year reality? A cruel joke. And the 30-month stay? A legal chokehold disguised as due process. We don’t need reform. We need arson.
Tildi Fletes
March 3 2026
It is critical to distinguish between patent term and regulatory exclusivity, as the article correctly notes. The U.S. Patent and Trademark Office (USPTO) and the Food and Drug Administration (FDA) operate under distinct statutory frameworks. Patent term is governed by 35 U.S.C. § 154, while regulatory exclusivities derive from 21 U.S.C. § 355. These are non-interchangeable mechanisms. For example, NCE exclusivity (5 years) is triggered upon approval, regardless of patent status. A drug may have expired patents but remain protected by exclusivity-this is not a loophole, it is statutory intent. The Orange Book listing is merely a transparency mechanism, not a legal barrier.
Siri Elena
March 4 2026
Oh, so now we’re shocked that pharma companies are, like, *businesses*? Who knew? 🤡 They’re not out here saving kittens-they’re out here maximizing shareholder value. And guess what? The system *lets* them. Every secondary patent, every 30-month stay, every pediatric extension? That’s not corruption. That’s capitalism with a PhD. If you want affordable drugs, stop pretending this is a moral issue and start treating it like the economic war it is. Also, congrats on writing a 3,000-word essay on why rich people are good at being rich.
Divya Mallick
March 4 2026
While the U.S. system is undeniably flawed, the global context is even more damning. India, China, and Brazil have long ignored secondary patents under TRIPS flexibilities-yet the West continues to act as if patent law is sacred scripture. This isn’t about innovation-it’s about imperial economic hegemony. The 20-year patent myth is weaponized to suppress generic manufacturing in the Global South. We’re not just talking about drug prices-we’re talking about millions of lives lost because a Western legal framework refuses to adapt. The Hatch-Waxman Act? A colonial relic dressed in legal jargon.
Alex Brad
March 6 2026
Patents start early. Clinical trials take years. Exclusivity is shorter than you think. Secondary patents delay generics. Regulatory exclusivity stacks on top. That’s it.
Dean Jones
March 6 2026
What fascinates me here isn’t just the mechanics of patent law, but the philosophical undercurrent: we’ve constructed a system where the value of human life is indirectly priced by the duration of a legal monopoly. The 20-year patent was never meant to be a ceiling-it was meant to be a starting point for a social contract between innovation and public good. But today, that contract has been hollowed out. The original intent of Hatch-Waxman was to balance two sacred values: the right to incentivize discovery and the right to access healing. Now, we’ve replaced that balance with a calculus of profit maximization. Every secondary patent filed isn’t just a legal maneuver-it’s a quiet surrender of our collective moral imagination. We’ve turned medicine into a derivative asset class, and patients into collateral.
Betsy Silverman
March 7 2026
I appreciate how thoroughly this breakdown is laid out. It’s easy to get lost in the jargon, but the distinction between patent term, regulatory exclusivity, and evergreening is crucial for anyone trying to understand why drug prices behave the way they do. The fact that a drug can have 30 patents but only one active ingredient is wild-and it’s not an accident. It’s strategy. And honestly, the most alarming part isn’t even the legal tactics-it’s how normalized they’ve become. We’ve stopped asking if this is fair, and started asking if it’s profitable. That shift is what we need to confront.